Using inert small molecules to control neuronal activity with DREADDs
Contributed by:
JUSTIN ENGLISH, ROTH LAB, Postdoctoral Fellow in the Roth Lab
Editor's note: This site is a collaboration with a Benchling user, Dr. Justin English, who is a postdoctoral researcher in Dr. Bryan Roth's laboratoryat the University of North Carolina at Chapel Hill. In this post he explains how to use the chemogenetic platform "DREADDs" to modify neuronal activity. If you want a similar website for your research or publication, let us know.
How does a brain work? One powerful way to address this question is to observe the relationship between neuronal activity and behavioral changes (e.g. movement, eating, or learning) in freely moving animals. In this guide, Dr. Justin English, a post-doctoral fellow of Bryan Roth's laboratory, joins Benchling to explain how neuroscientists are using the chemogenetic technology they have developed - termed "DREADDs" - to understand the animal brain.
DREADDs are an open source academic resource. As such, the Roth laboratory has also made available with this guide all plasmids and viral vectors for DREADD-based research. See links to these at the end of this post!
How do you control neuronal activity with chemogenetics?
Neurons communicate with each other through chemical signals, known as neurotransmitters. These neurotransmitters can bind to membrane receptors on receiving neurons and activate signaling pathways to modify the activity of neurons. Most membrane receptors are G protein-coupled receptors (GPCRs). Common neurotransmitters of the brain, such as dopamine and serotonin, bind to GPCRs to modulate neuronal activities.
In traditional methods to perturb neuronal function, scientists use drugs that target GPCRs and other membrane proteins. By activating or silencing specific receptors, researchers can modulate neuronal activity to understand how the brain operates. However, the drugs used to target receptors are often nonspecific, affecting the activity of more than one GPCR. Furthermore, the same GPCR is often present in multiple tissues, meaning that even an exquisitely specific drug can activate GPCRs across the brain and body. This multi-site expression can confound interpretation of even the best drug studies.
To overcome this problem, the Roth Laboratory engineers GPCRs so that they only respond to a specific biologically inert chemical (a designer drug). We call these modified GPCRs Designer Receptors Exclusively Activated by Designer Drugs (DREADDs). DREADD-free neurons are unresponsive to the designer drug while cells expressing the DREADD respond robustly to low concentrations of the designer molecule.
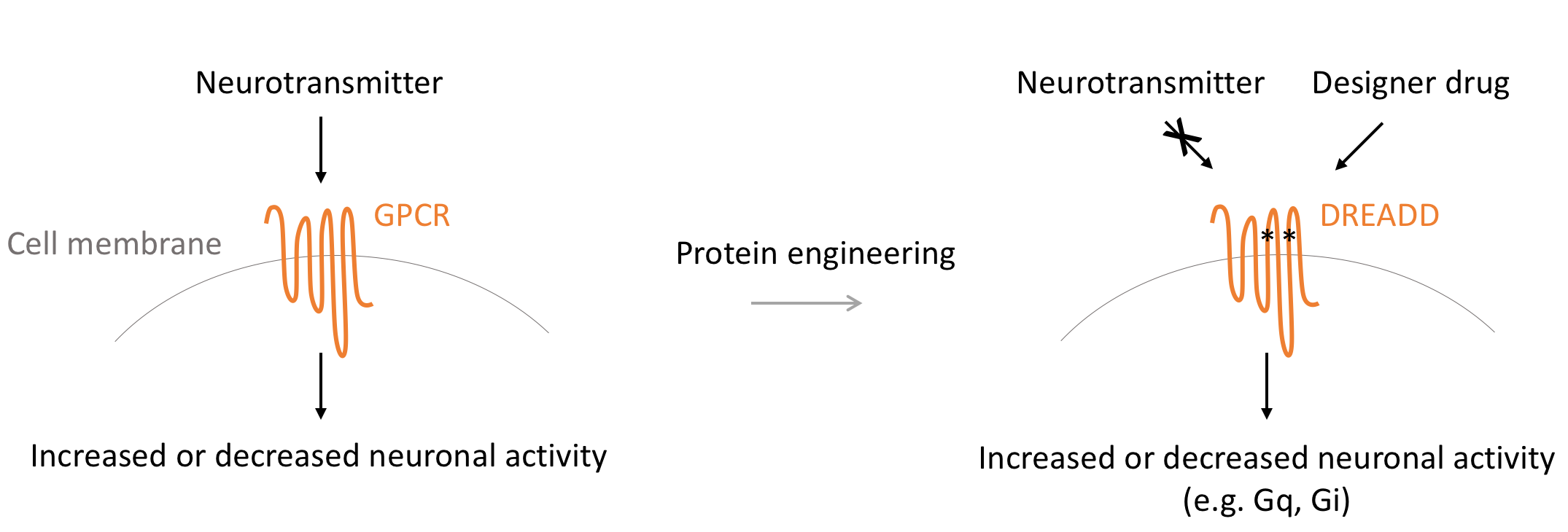
What are the advantages of DREADDs?
There are several benefits of using DREADDs to understand the brain:
You can control neuronal activity non-invasively:You can deliver the designer drug in the peripheral system, and the drug will cross the blood-brain barrier to activate DREADD-expressing neurons.
You can select which signaling pathways in neurons to control:Since DREADDs are modified from GPCRs, based on the DREADD you choose, you can precisely control Gq, Gi or Gs-signaling pathways in cells.
DREADDs and the corresponding designer drugs are non-toxic:You can express DREADDs and use the corresponding designer drug to manipulate the activity of neurons over days or months while keeping the neurons healthy1.
When should I use DREADDs instead of other neuromodulatory tools?
You should always consider the nature of your research when choosing your experimental toolset. Like all methods, DREADDs have their own unique advantages and limitations. DREADDs are an excellent choice if you need to:
Chronically modulate neuronal activity:DREADDs can remain active for several hours following administration of a single dose of the corresponding designer drug. The drug can also be administered repeatedly to enable multi-day or chronic modulation of neuronal activity.
Experiment without instrumentation:DREADDs require no cemented or anchored instrumentation to operate. The drug can be provided through injections or water supply, allowing test animals to roam freely, perform complex tasks, and socialize.
Generate interventions using endogenous signaling pathways:DREADDs are minimally modified forms of endogenous GPCRs and are therefore directly coupled to their original signaling pathway.
hM3Dq
Click herefor more information about the plasmids for hM3Dq.
hM3Dq is a modified form of the human M3 muscarinic (hM3) receptor. It can be activated by the inert clozapine metabolite clozapine-N-oxide (CNO), engaging the Gq signaling pathway. Gq signaling releases intracellular calcium stores and enhances neuronal excitation. Thus, neurons expressing hM3Dq treated with CNO are observed to have dramatically increased firing rates2.
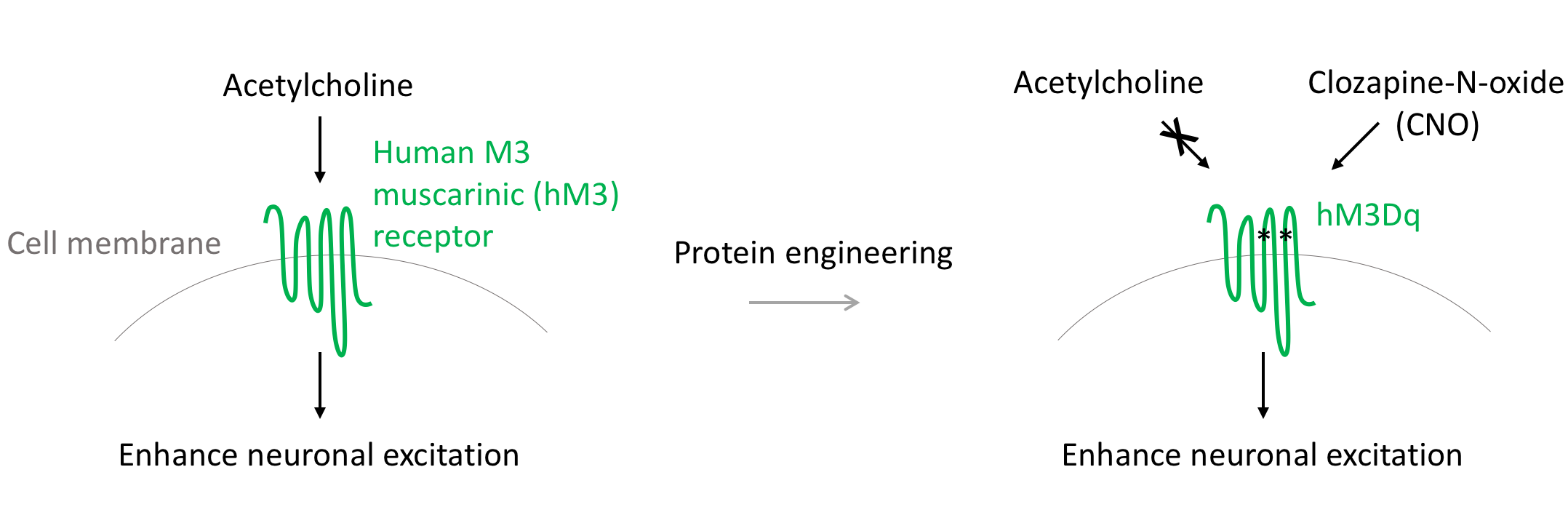
Researchers have used hM3Dq to increase the firing rate of neurons of interest. For example, Krashes et al. used the hM3Dq to test the hypothesis that AgRP neurons promote feeding and weight gain in mice. The researchers expressed hM3Dq in AgRP neurons in mice and used CNO to activate the receptors. The firing rate of hM3Dq-expressing neurons increased, leading the mice to eat more, move less, and accumulate fat, confirming the hypothesis3.
hM4Di
Click herefor more information about the plasmids for hM4Di.
hM4Di is a modified form of the human M4 muscarinic (hM4) receptor. It can be activated by the inert clozapine metabolite clozapine-N-oxide (CNO), engaging the Gi signaling pathway. Gi signaling in neurons opens potassium channels resulting in an influx of potassium ions that decreases the resting membrane potential of neurons and their capacity to depolarize. Thus, neurons expressing hM4Di treated with CNO are observed to have dramatically decreased firing rates2.
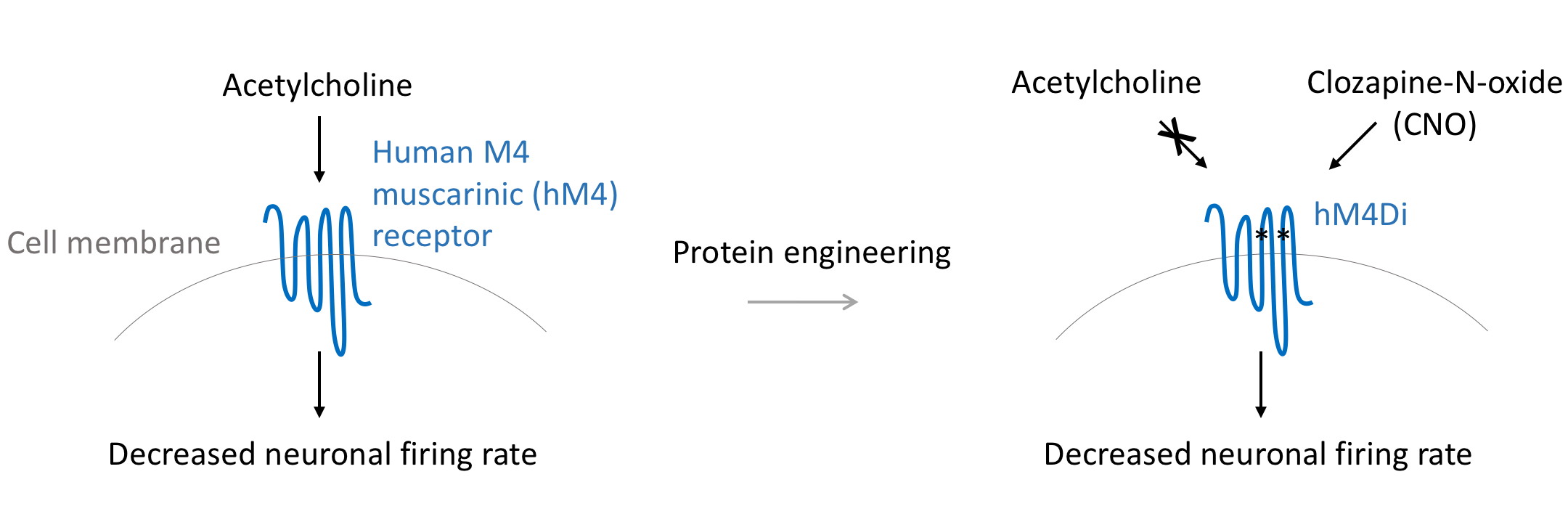
Researchers have used hM4Di to reduce the firing rate of neurons of interest. For example, orexin neurons play an important role to control sleep and wakefulness. Sasaki et al. used CNO to suppress the activity of hM4Di-expressing orexin neurons and the animal spent less time awake4.
KORD
Click herefor more information about the plasmids for KORD.
KORD is a modified form of the human kappa opioid (KOR) receptor. KORD is activated by the inert compound salvinorin B (SALB), resulting in activation of Gi signaling pathway. As with hM4Di, Gi signaling greatly decreases neuronal firing. Therefore, neurons expressing KORD are observed to have dramatically decreased firing rates5.
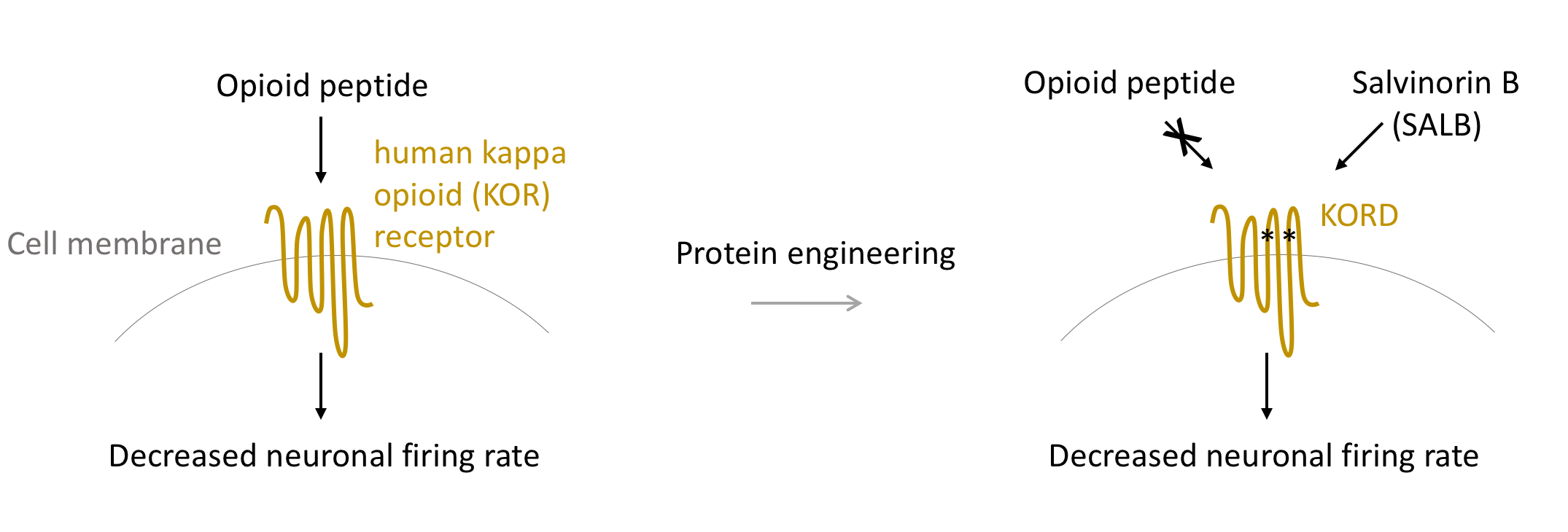
Researchers have co-expressed KORD and hM3Dq within the same neuronal population to control neuronal activity bi-directionally. For example, Vardy et al. expressed both hM3Dq and KORD to the neurons that control movement (VTA). Exciting these neurons with CNO makes the animals move less, and inhibiting these neurons with SALB enhanced movement of the animals5.
For a comprehensive overview of DREADDs, see Roth, Neuron, 2016.
References
Eldridge, Mark AG et al."Chemogenetic disconnection of monkey orbitofrontal and rhinal cortex reversibly disrupts reward value." Nature Neuroscience(2015).
Armbruster, Blaine N et al."Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand." Proceedings of the National Academy of Sciences(2007).
Krashes, Michael J et al."Rapid, reversible activation of AgRP neurons drives feeding behavior in mice." The Journal of Clinical Investigation(2011).
Sasaki, Koh et al."Pharmacogenetic modulation of orexin neurons alters sleep/wakefulness states in mice." PLoS One(2011).
Vardy, Eyal et al."A new DREADD facilitates the multiplexed chemogenetic interrogation of behavior." Neuron(2015).